Quantitative determination using gas chromatography
Objectives
Here is an Example of a Lab Report in Chemistry. There are three main objectives of this experiment:
- To conduct a quantitative determination using gas chromatography
- To estimate the maintenance time for hexane and ethanol
- To study the reaction between tR and atomic carbon in homologous series
Introduction
Graph chromatography is a chromatography technique that is used in analytical chemistry mainly for separating and analyzing compounds (Pavia, Gary, Kritz, & Engel, 2006). It is used for analysis of compounds that can be vaporizing without decomposing. Example of a Lab Report in Chemistry It is used for testing the purity of substances or separating different components that are mixed, where it can also determine the relative amounts of each compound in a mixture. In gas chromatography, the mobile phase has a carrier gas, which is an inert gas like helium or unreactive gas like hydrogen. Station phase uses liquid or polymer adsorbed on the surface as shown below:
There are different locations of gas chromatography. These include warm conductive detectors (TCD), electron –capture detectors (ECD), Thermionic detectors (TID), and fire ionization detectors (FID). In this experience, FID was used as the main locator. Example of a Lab Report in Chemistry
Example of a Lab Report in Chemistry Procedure
There were five main steps that were followed in this test as explained below:
First, sample was drawn by putting the needle into the liquid sample and polluting the plunger. The accurate amount of sample was drawn. About 2/il of sample was drawn using a 10/il syringe. The plunger was slowly and carefully put back ensuring that not sample was left on the needle.
Second, the syringe was held in two hands as shown in the figure below.
Third, the syringe was brought to the same level with the injection port. Holding the needle straight, it was let to touch the center of the septum. Example of a Lab Report in Chemistry
Fourth, without injecting, the need was pushed through the septum.
Lastly, the plunger was pushed slowly to inject the sample and the syringe was pulled out of the septum and the injection port. Example of a Lab Report in Chemistry
Example of a Lab Report in Chemistry Results
The following results were obtained:
Figure 1: ETHANOL peak. Time: 15 min. Level: 1058 uV
This graph shows that ethanol peak at 15 minues and 1058uV
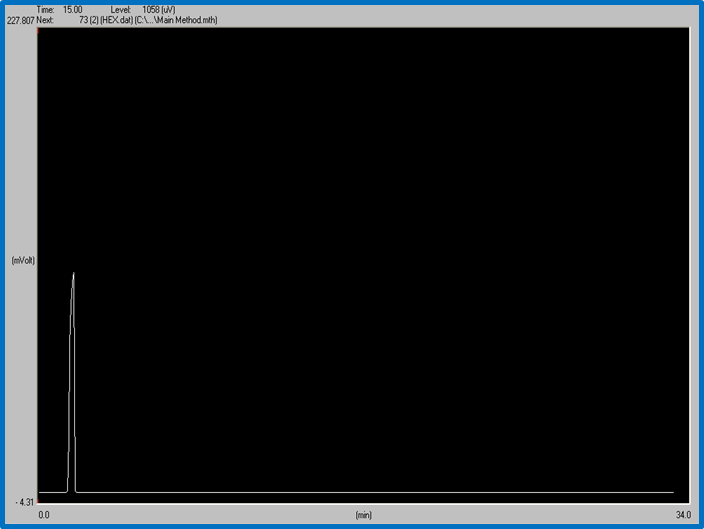
Figure 2: HEXANE shows higher peak. Time: 27.85 min. Level:1052 uV Example of a Lab Report in Chemistry
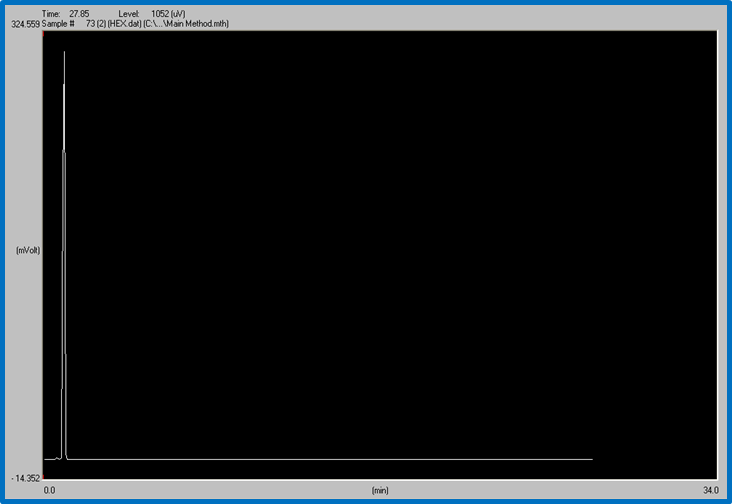
As shown in the figure, Hexane peaked faster than ethanol at 27.85 minutes and 1052 uV.
Figure 3: MIX of ethanol and hexane shows the two peaks on the same graph. Time: 19 min. Level: 1050 uV
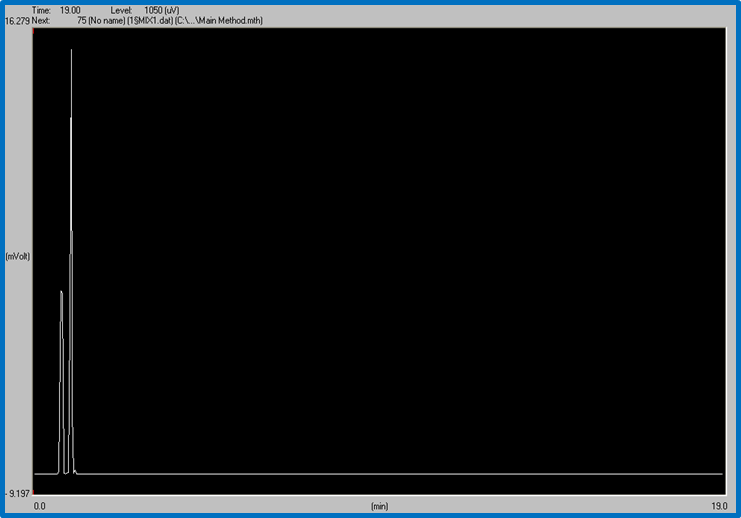
As shown in the figure, a mixture of both ethanol and hexane peaked highest at 19min and 1050 uV.
Discussion
Example of a Lab Report in Chemistry This experiment was mainly estimating the maintenance time for hexane, ethanol, and a mixture of both. First, ethanol was infused, then hexane, and lastly a mixture of both. Gas chromatography has a non-polar stationary stage. Ethanol is polar, which means when infused, there was polarity between the two. This is what made ethanol to peak faster at 15 minutes compared to the hexane. Comparatively hexane is non-polar and it invests more energy in leaving the column compared to ethanol, peaking at 27 minutes. Because the column in gas chromatography non-polar, ethanol peaks first and then hexane last, remaining in the column for a longer time. The result is a longer peak for ethanol and shorter for hexane. Example of a Lab Report in Chemistry
References
Pavia, D.L., Gary, M. L., Kritz, G.S., & Engel, R.G. (2006). Introduction to Organic Laboratory Techniques (4th Ed.). Thomson Brooks/Cole.
DO YOU NEED A LAB REPORT LIKE THIS? ORDER NOW
